HPLC Method for Separation of 1-Butylpyridinium chloride and 1-Ethylpyridinium bromide on Newcrom AH Column
Separation type: Liquid Chromatography Mixed-mode
1-Butylpyridinium chloride and 1-Ethylpyridinium bromide are types of ionic liquids. Ionic liquids are salts in which the ions are poorly coordinated, which results in these solvents being liquid below 100°C, or even at room temperature (ambient temperature). At least one ion has a delocalized charge and one component is organic, which prevents the formation of a stable crystal lattice.
1-Butylpyridinium chloride has a butyl group attached to a pyridinium ring, and its counterion is chloride (Cl-).
1-Ethylpyridinium bromide, on the other hand, has an ethyl group attached to a pyridinium ring, and its counterion is bromide (Br-).
These compounds are part of a larger class of pyridinium-based ionic liquids. They have various applications, including in electrochemistry, organic synthesis, and as solvents for polymers. Like other ionic liquids, their properties (such as melting point, viscosity, and solvation potential) can be adjusted by selecting different counterions or changing the groups attached to the pyridinium ring.
1-Butylpyridinium chloride and 1-Ethylpyridinium bromide,” belong to a class of compounds known as pyridinium salts. These compounds are derived from the parent compound pyridine, which is a six-membered aromatic ring containing one nitrogen atom.
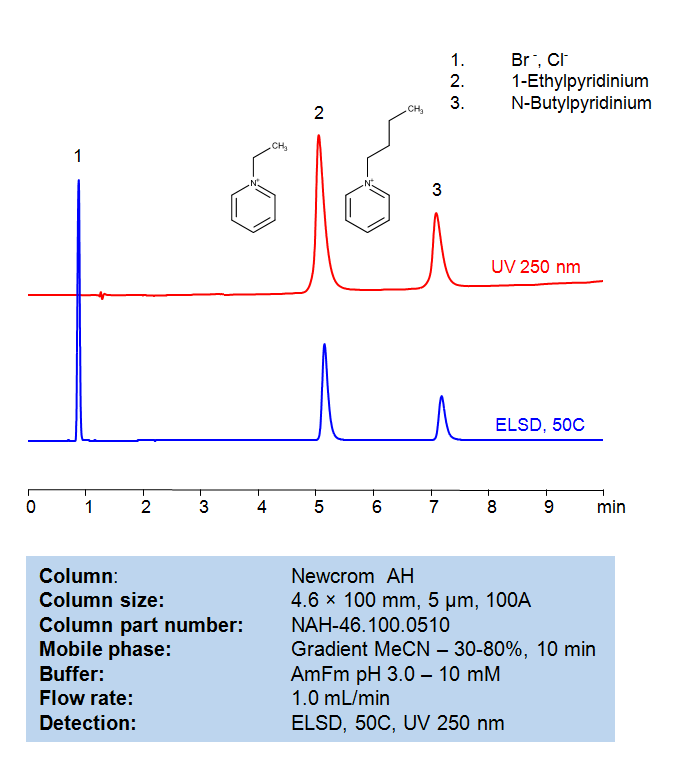
Methodenparameter
| Column | Newcrom AH, 4.6 x 100 mm, 5 µm, 100 Å, dual ended |
|---|---|
| Mobile Phase | Gradient MeCN/H2O – 30-80%, 10 min |
| Buffer | AmFm pH 3.0- 10 mM |
| Flow Rate | 1.0 mL/min |
| Detection | ELSD, the nebulizer and evaporator temperatures 50 °C, with a gas flow rate of 1.6 Standard Liters per Minute (SLM) (MS- compatible mobile phase) |
| Limit of Detection | UV- 30 ppb, ELSD – 70 ppb |
| Injection Volume | 1µl |