Determination Imatinib Mesylate on Obelisc R Column
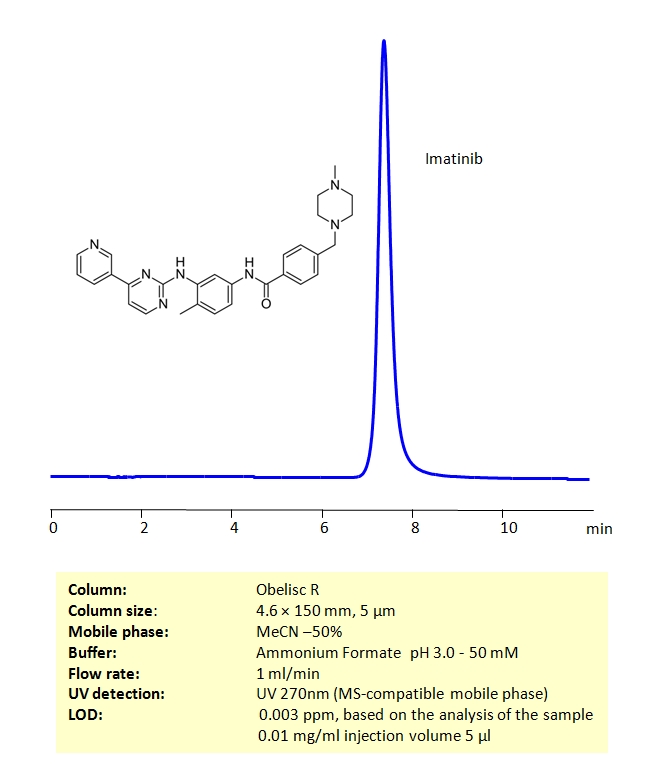
High Performance Liquid Chromatography (HPLC) Method for Analysis of Imatinib mesylate .
Imatinib mesylate is a classified as a signal transduction inhibitor with the chemical formula C29H31N7O . It is an oral targeted therapy medication that is effective in treating chronic myeloid leukemia and metastatic gastrointestinal stromal tumors. Commercial versions of the drug were a topic of controversy when cancer specialists published a letter condemning the high costs of cancer treatments.
Imatinib mesylate can be retained on an Obelisc R column, which has both positive and negative ion-pairs embedded in the stationary phase, allowing for the fine tuning and separation of a wide range of compounds with different ionic properties. Imatinib mesylate can be determined isocratically using a simple MS-compatible mobile phase of acetonitrile (ACN) and water with Ammonium Formate (AmFm) buffer and detected by UV at 270 nm, ELSD, CAD or LC/MS.
Methodenparameter
| Column | Obelisc R, 4.6 x 150 mm, 5 µm, 100 Å, dual ended |
|---|---|
| Mobile Phase | MeCN -50% |
| Buffer | Ammonium Formate pH 3.0 – 50 mM |
| Flow Rate | 1.0 mL/min |
| Detection | UV 270 nm |