HPLC Method for Separation of Michler’s ketone and Crystal Violet on Primesep 100 Column
https://sielc.com/hplc-determination-of-crystal-violet-michlers-ketoneChromatogram
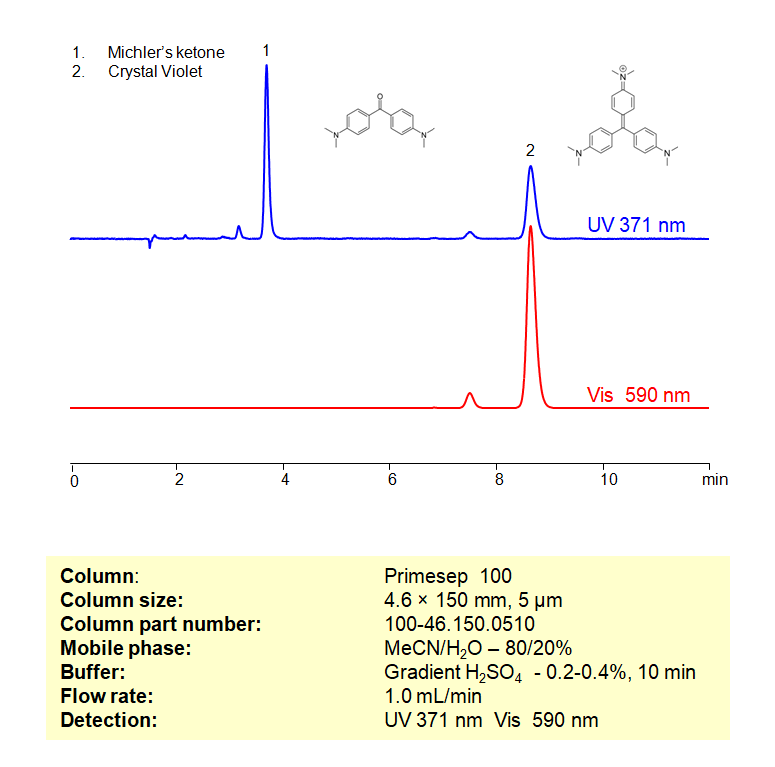
Description
· Separation type: Liquid Chromatography Mixed-mode
HPLC Method for Analysis of Michler’s ketone , Crystal Violet on Primesep 100 Column by SIELC Technologies
Michler’s ketone (4,4′-bis(dimethylamino)benzophenone) is a chemical compound used primarily in the synthesis of dyes and pigments. It has the chemical formula C17H17N3O2. The compound is named after the German chemist Viktor Michler, who first prepared it.
Michler’s ketone is synthesized by condensing two equivalents of dimethylaniline with one equivalent of phosgene. The reaction involves the formation of a carbonyl group (C=O) that links two dimethylaniline moieties. The resulting structure features two aromatic rings (benzene rings) connected by a carbonyl group, with dimethylamino groups attached to each aromatic ring.
Michler’s ketone is a valuable intermediate in the dye industry, where it is used to prepare various triarylmethane dyes, including Malachite Green, Methyl Violet, and Crystal Violet. The strong electron-donating properties of the dimethylamino groups make it a useful compound for synthesizing dyes with vibrant and stable colors.
Michler’s ketone and Crystal Violet can be separated, retained, and analyzed on a Primesep 100 mixed-mode stationary phase column using an isocratic analytical method with a simple mobile phase of water, Acetonitrile (MeCN), and a sulfuric acid (H2SO4) buffer. This analysis method can be detected in the UV-Vis regime at 540, 590, and 200 nm.
Method Parameters
| Mobile Phase | MeCN/H2O – 80/20% |
|---|---|
| Buffer | Gr H3PO4 – 0.2-0.4%, 10 min |
| Flow Rate | 1.0 ml/min |
| Detection | UV 371, Vis 590 nm |
| Peak Retention Time | 3.71, 8.57 min |
| Class of Compounds | Dyes |
| Analyzing Compounds | Michler’s ketone,Crystal Violet |