HPLC MS Method for Analysis of Trichloroacetic acid (TCA) on BIST A+ Column
Separation type: Liquid Chromatography Reversed-phase
Trichloroacetic acid (TCA) is a chemical compound with the formula CCl3COOH. It is a strong organic acid and a derivative of acetic acid, where three of the four hydrogen atoms in the methyl group of acetic acid are replaced by chlorine atoms.
Trichloroacetic acid is commonly used in various applications, including:
Chemical Synthesis: TCA serves as a reagent in various chemical reactions, such as the production of pharmaceuticals, dyes, and pesticides.
Protein Precipitation: In biochemistry and molecular biology, TCA is often used for the precipitation of proteins from solution. It is particularly useful for removing proteins from samples for further analysis.
Skin Care: TCA is also used in dermatology as a peeling agent to treat various skin conditions, including acne scars, sun damage, and wrinkles. It promotes exfoliation and the regeneration of skin cells.
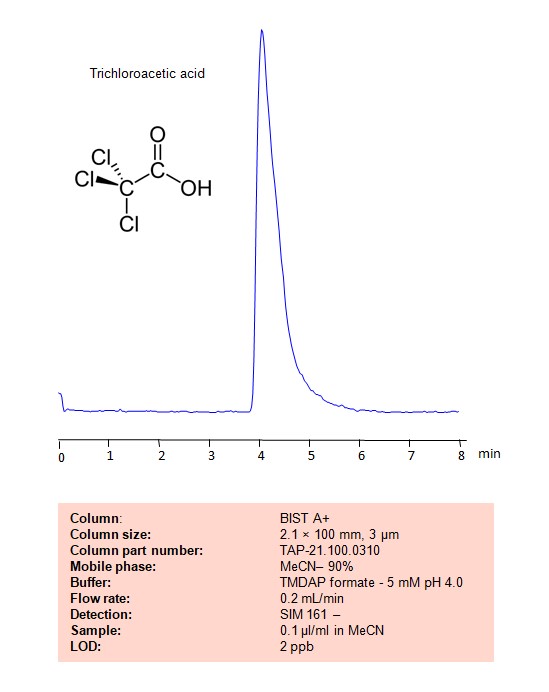
Methodenparameter
| Column | BIST A+, 2.1 x 100 mm, 3 µm, 100 Å, dual ended |
|---|---|
| Mobile Phase | MeCN/H2O – 90/10% |
| Buffer | TMDAP formate – 5 mM pH 4.0 |
| Flow Rate | 0.2 mL/min |
| Detection | SIM161- |
| Sample | 0.1µl/ml |