HPLC Method for Separation of Methyl Red and Ethyl Red on Primesep 100 Column
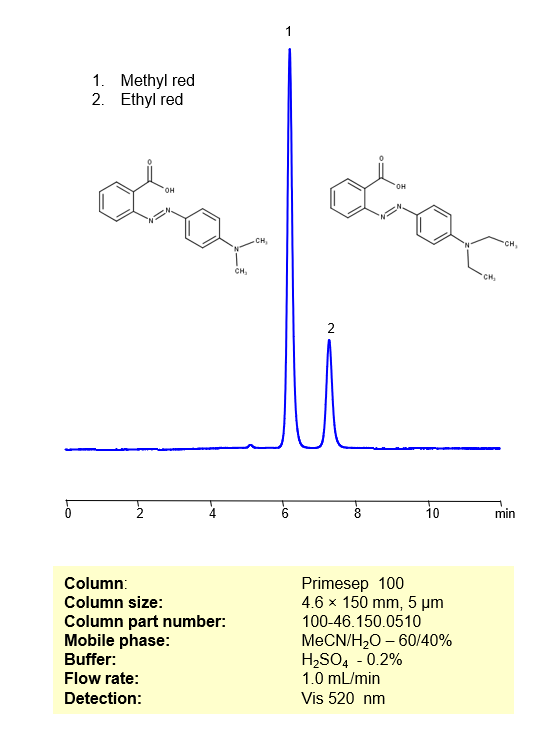
Methyl Red , also known as C.I. Acid Red 2 , is a dark-red, single-charged basic dye that turns a deep red in acidic (low pH) solutions and yellow at solution with pH above 6.2. It has the chemical formula C15H15N3O2 . You can find detailed UV spectra of Methyl Red and information about its various lambda maxima by visiting the following link.
Ethyl red is a pH indicator with C17H19N3O2as its molecular structure. When the pH transitions from acidic to neutral, Ethyl red turns from yellow to red, hence the name. Outside of experiments, it is occasionally used as a dye in textiles and foods. You can find detailed UV spectra of Ethyl Red and information about its various lambda maxima by visiting the following link.
Methyl Red , Ethyl red can be separated, retained, and analyzed on a Primesep 100 mixed-mode stationary phase column using an isocratic analytical method with a simple mobile phase of water, Acetonitrile (MeCN), and a sulfuric acid (H2SO4) buffer. This analysis method can be detected in the Visible spectrum at 520 nm.
Methodenparameter
| Column | Primesep 100, 4.6 x 150 mm, 5 µm, 100 Å, dual ended |
|---|---|
| Mobile Phase | MeCN/H2O – 60/40% |
| Buffer | H3PO4 – 0.2% |
| Flow Rate | 1.0 mL/min |
| Detection | Vis, 520 nm |