HPLC Analysis of Active Drug in a Formulation
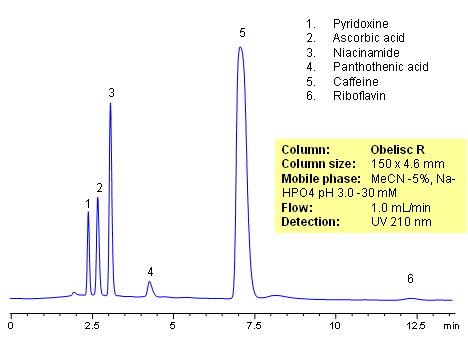
HPLC method for separation of active ingredients of drug/supplemental composition was developed on an Obelisc R trimodal HPLC column. Compounds are retained by combination of reversed-phase, cation-exchange and anion-exchange mechanisms. Compounds are well separated, and method can be used for quantitation of pyridoxine, ascorbic acid, niacinamide, pantothenic acid, caffeine and riboflavin in a mixture or as separate compounds in various complex mixtures. Various detection techniques can be applied for quantitation (ELSD, UV, LC/MS, Corona). This HPLC method can be adopted as general approach for analysis of active drug components in various formulations.
SIELC has developed the Obelisc™ columns, which are mixed-mode and utilize Liquid Separation Cell technology (LiSC™). These cost-effective columns are the first of their kind to be commercially available and can replace multiple HPLC columns, including reversed-phase (RP), AQ-type reversed-phase, polar-embedded group RP columns, normal-phase, cation-exchange, anion-exchange, ion-exclusion, and HILIC (Hydrophilic Interaction Liquid Chromatography) columns. By controlling just three orthogonal method parameters - buffer concentration, buffer pH, and organic modifier concentration - users can adjust the column properties with pinpoint precision to separate complex mixtures.
Methodenparameter
| Column | Obelisc R , 4.6×150 mm, 5 µm, 100 Å |
|---|---|
| Mobile Phase | MeCN/H2O -5/95% |
| Buffer | NaHPO4pH 3.0 – 30 mM |
| Flow Rate | 1.0 mL/min |
| Detection | UV, 210 nm |