HPLC Separation of Caffeine, 3- Methylxanthine, 1- Methylxanthine, Xanthine
https://sielc.com/separation_of_caffiene_3-methylxanthine_1-methylxanthine_xanthineChromatogram
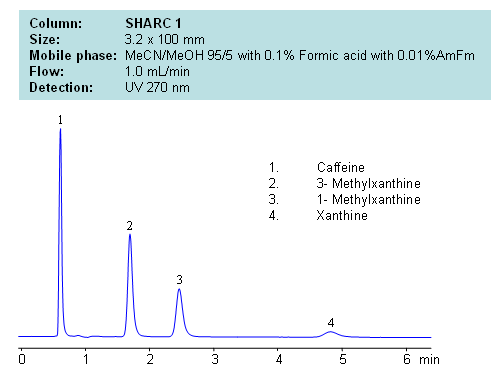
HPLC Separation of Caffeine, 3- Methylxanthine, 1- Methylxanthine, Xanthine
Description
Application Notes: Xanthines are polar neutral compounds which are hard to retain and separate by traditional reversed-phase chromatography. However a hydrogen bonding method makes separation possible due to an observable correlation between the number of hydrogens available for interaction and retention time. Molecules with no hydrogens available for interactions retain less, and compound with multiple hydrogen donors retain the most. Retention time can be controlled by changing ratio of ACN:MeOH. Other protic and aprotic solvents can be used to control retention time and selectivity of separation.
Application Columns: SHARC 1, 3.2×100 mm, 5 um, 100A, To learn more about SHARC 1 columns click here . To order this column click here . To see more chromatographic separations check our web site.
Application Compounds: Caffeine, 3-methylxanthine, 1-methylxanthine, and xanthine
Method Parameters
| Mobile Phase | MeCN/MeOH |
|---|---|
| Buffer | AmFm, Formic acid |
| Flow Rate | 1.0 ml/min |
| Detection | UV, 270 nm |
| Class of Compounds | Drug, Acid, Hydrophilic, Ionizable, Vitamin, Supplements |
| Analyzing Compounds | Caffeine, 3- Methylxanthine, 1- Methylxanthine, Xanthine |