HPLC Separation of Haloaromatics and Quinones on Newcrom B Column
https://sielc.com/hplc-separation-of-haloaromatics-and-quinones-on-newcrom-b-columnChromatogram
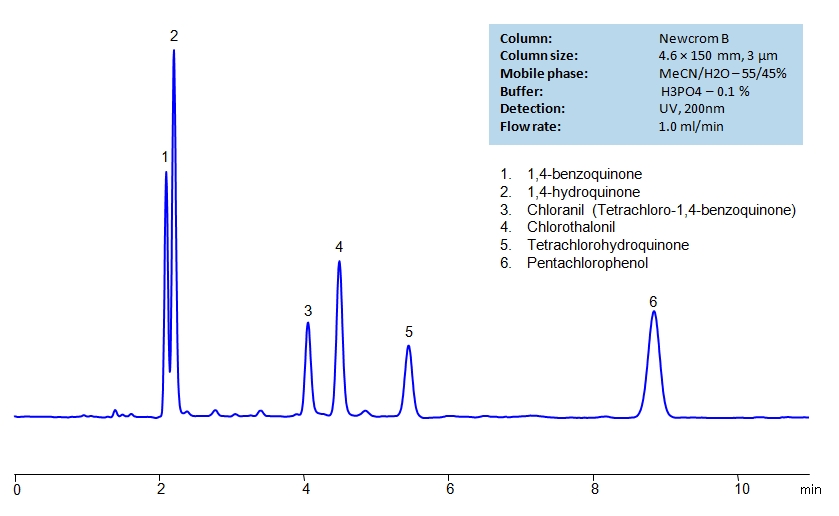
Description
High Performance Liquid Chromatography (HPLC) Method for Analysis of Benzoquinone , Hydroquinone , Chlorothalonil , Chloranil , Pentachlorophenol , Tetrachlorohydroquinone .
1,4-benzoquinone , also known as para -quinone, is a compound with the formula C 6 H 4 O 2 . It is typically used as a precursor to hydroquinone, which is a reducing agent and antioxidant. It also serves as a dehydrogenation reagent and dienophile in Diels-Alder reactions.
1,4-hydroquinone is an aromatic derivative of benzene with the chemical formula C 6 H 6 O 2 . It is often used in skin whitening, although it has been banned by the United States Food and Drug Administration for over-the-counter use due to being a potential carcinogen. It can cause a variety of disease including but not limited to ochronosis. thyroid follicular cell hyperplasias, mononuclear cell leukemia, and adenomas. Agencies across the world encourage research into other agents to treat hyperpigmentation. You can find detailed UV spectra of hydroquinone and information about its various lambda maxima by visiting the following link.
Chloranil , also known as tetrachloro-1,4-benzoquinone, is a quinone with the chemical formula C 6 Cl 4 O 2 . It is a planar molecule that functions as an oxidant. It serves as a hydrogen acceptor and is more electrophilic than quinone. Chloranil is used to test for free secondary amines, which is useful to check for the presence of proline derivatives. Commercially, it is a precursor to dyes.
Chlorothalonil is a compound with a variety of uses as a fungicide, wood protectant, pesticide, and acaricide. It is used predominantly on peanuts, potatoes, and tomatoes. Outside of agriculture, it is also used in paints, resins, emulsions, and coatings. It’s chemical formula is C 8 Cl 4 N 2 .
Tetrachlorohydroquinone , also known as TCHQ , is a chlorinated organic compound with the chemical formula C 6 H 2 Cl 4 O 2 . It is a metabolote of the biocide pentachlorophenol. It causes damage to cells by increasing reactive oxygen species (ROS). It is harmful if swallowed and can cause serious eye damage.
Pentachlorophenol is a manufactured chemical with the chemical formula C 6 HCl 5 O . It is most often used as herbicide, insecticide, fungicide, algaecide, and disinfectant. Exposure to it can cause damage to liver, kidney, blood, lungs, eyes, skin, and mouth. It is classified as a probable human carcinogen.
Method Parameters
| Mobile Phase | MeCN/H2O – 55/45% |
|---|---|
| Buffer | H3PO4 – 0.1% |
| Flow Rate | 1.0 ml/min |
| Detection | UV 200 nm |
| Class of Compounds | Haloaromatics, Quinones, Hydrophobic |
| Analyzing Compounds | Benzoquinone,Hydroquinone,Chlorothalonil,Chloranil,Pentachlorophenol,Tetrachlorohydroquinone |