HPLC Method for Analysis mixture of Xanthines and Uric Acid BIST B+ by SIELC Technologies
https://sielc.com/hplc-method-uric-acid-and-xanthinesChromatogram
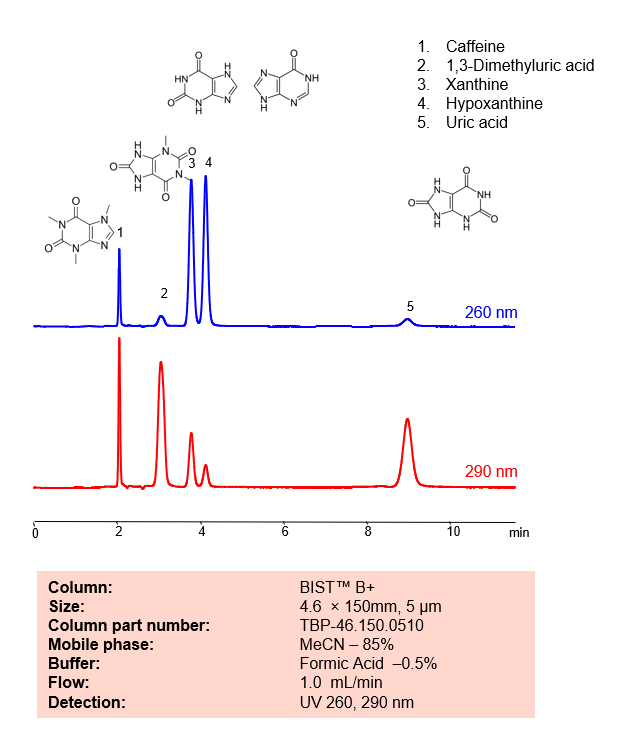
Description
Xanthines and uric acid are related compounds in the body and both are involved in the metabolism of purines.
Xanthines are a group of alkaloids that are widely distributed in plants, and also occur in the tissues and fluids of animals. They are known to stimulate the central nervous system and cardiac muscle, and also have diuretic effects.
In the body, xanthines are intermediates in the degradation of adenosine monophosphate to uric acid. This metabolic pathway starts with adenosine monophosphate (AMP), which is deaminated to form inosine monophosphate (IMP). IMP is then converted into a xanthine known as hypoxanthine. Hypoxanthine is then oxidized to xanthine, and finally, xanthine is further oxidized to uric acid. Both of the oxidation steps are catalyzed by the enzyme xanthine oxidase.
Caffeine is a natural stimulant and methylxanthine alkaloid. with the molecular formula C 6 H 10 N 4 O 2 . Caffeine can be found in a variety of plants, including tea, coffea, cocoa, kola nuts, and guarana. Ingestion of it can increase alertness and cognitive function. It can also cause worsening anxiety, heart palpitations, and headaches You can find detailed UV spectra of caffeine and information about its various lambda maxima by visiting the following link.
1,3-Dimethyluric acid is an oxopurine with the chemical formula C 7 H 8 N 4 O 3 . It is a is a metabolite of caffeine and theophylline with antioxidant properties. It is used as a biomarker in urine for activity of the CYP1A2 enzyme.
Xanthine is a purine base with the chemical formula C 5 H 4 N 4 O 2 . It is a product on the pathway of purine degradation. Numerous stimulants are derived from xanthine.
Hypoxanthine is a naturally occurring purine derivative with the chemical formula C 5 H 4 N 4 O . It is a reaction intermediate in the metabolism of adenosine as well as a metabolite found in Escherichia coli.
Method Parameters
| Mobile Phase | MeCN – 85% |
|---|---|
| Buffer | FA – 0.5% |
| Flow Rate | 1.0 ml/min |
| Detection | UV 260, 290 nm |
| Peak Retention Time | 2.01, 3.02, 4.2, 9.09 min |
| Class of Compounds | Acid, Xanthines |
| Analyzing Compounds | Caffeine,1,3-Dimethyluric acid,Xanthine,Hypoxanthine,Uric acid |