HPLC Determination of Nitrilotriacetic acid (NTA) and Ethylenediaminetetraacetic acid (EDTA) on Newcrom BH Column
https://sielc.com/hplc-determination-of-nitrilotriacetic-acid-nta-and-ethylenediaminetetraacetic-acid-edtaChromatogram
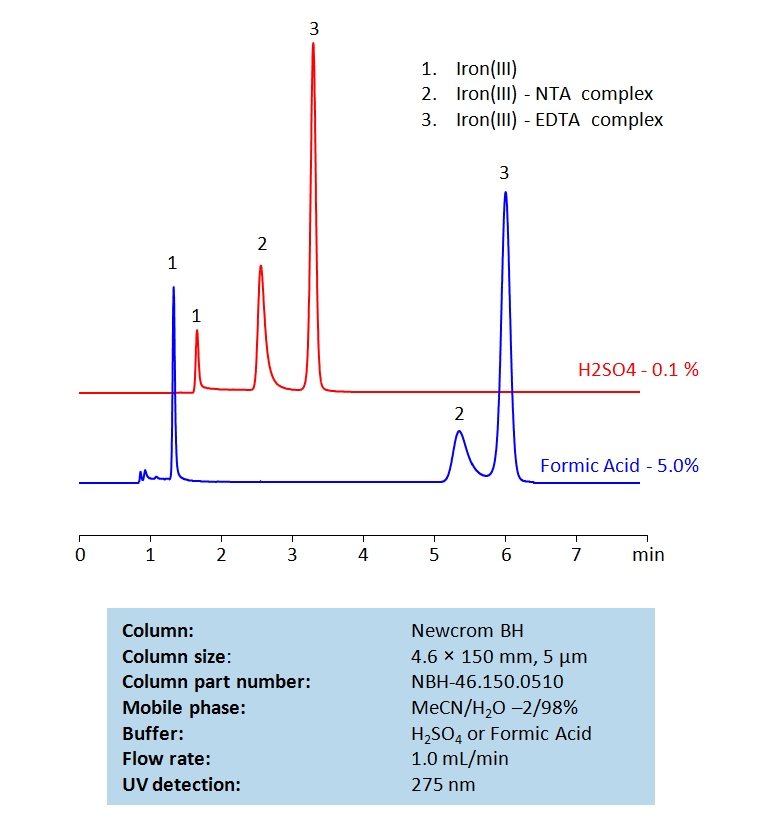
Description
High Performance Liquid Chromatography (HPLC) Method for Analysis of Nitrilotriacetic acid , EDTA (Ethylenediaminetetraacetic Acid) .
NTA Standards Solution A: For the preparation of the NTA standard solution, 5 mg of NTA was accurately weighed and transferred into a 5 mL volumetric flask and dissolved in water with sonication. The NTA stock solution (1 mg/mL) should be stored in a cold, dark place and can be used for upto a week to prepare standards of required concentration.
Nitrilotriacetic acid ( NTA ) and Ethylenediaminetetraacetic acid ( EDTA ) are two common chelating agents used often in laboratory and medical testing. They are often used to bind to, identify, and analyze metal ions. Below is a method we developed to retain and measure Fe (III) using NTA and EDTA complexes on a Newcrom BH mixed-mode column, with a mobile phase of acetonitrile (MeCN), water, and a buffer of either sulfuric acid (H2SO4) or formic acid:
EDTA Standards Solution B: For the preparation of the EDTA standard solution, 5 mg of EDTA was accurately weighed and transferred into a 5 mL volumetric flask and dissolved in 0.001N NaOH water solution with sonication or magnetic stirrer mixing. Filtered The EDTA stock solution (1.0 mg/mL) should be stored in a cold dark place and can be used for a week to prepare standards of required concentration. NTA Standard – Solution A: For the preparation of the NTA standard solution, 5 mg of NTA was accurately weighed and transferred into a 5 mL volumetric flask and dissolved in water with sonication. The NTA stock solution (1 mg/mL) should be stored in a cold, dark place and can be used for upto a week to prepare standards of required concentration.
EDTA Standard – Solution B: For the preparation of the EDTA standard solution, 5 mg of EDTA was accurately weighed and transferred into a 5 mL volumetric flask and dissolved in 0.001N NaOH water solution with sonication or magnetic stirrer mixing. Filter the EDTA stock solution (1.0 mg/mL) should be stored in a cold dark place and can be used for a week to prepare standards of required concentration.
Iron(III) chloride – Solution C: The standard stock solution of Iron(III) chloride (10 mg/ml) was prepared in water. 50 mg of FeCl3 was accurately weighed and transferred into a 5 mL volumetric flask and dissolved in water, with sonication if needed.
General procedure for Ferric NTA and EDTA complex analysis: To make a sample for analysis, mix 100 µL each Solution A and Solution B (or unknown sample) with 100 µL Solution C and 700 µL of water. Place this mixture in a plastic HPLC vial for analysis. Setup instrument and column according to the method provided.
Method Parameters
| Mobile Phase | MeCN/H2O – 2/98% |
|---|---|
| Buffer | H2SO4 or Formic Acid |
| Flow Rate | 1.0 ml/min |
| Detection | UV 275nm |
| Class of Compounds | Acid, Hydrophilic |
| Analyzing Compounds | Nitrilotriacetic acid,EDTA (Ethylenediaminetetraacetic Acid) |