USP Methods for the Analysis of an Analgesic Mixture Using the Legacy L1 Column
https://sielc.com/Application-USP-Methods-for-the-Analysis-of-an-Analgesic-MixtureChromatogram
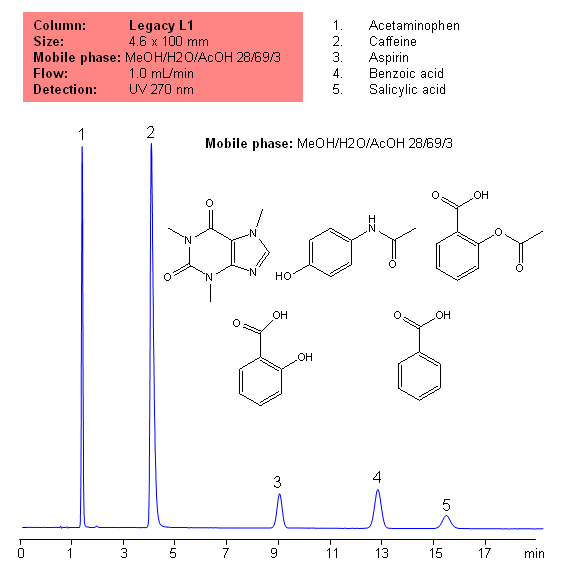
Description
Application Notes: Acetametaphin, aspirin, and caffeine tablets contain not less than 90 percent and not more than 110 percent of the labeled amounts if acetametaphin, asprin, and caffeine according the USP methods. USP HPLC method for separation of acetaminophen, aspirin and caffeine was developed on Legacy L1 column according to US Pharmacopeia methodology. L1 classification is assigned to reversed-phase HPLC column contains C18 ligands. Support for the material is a spherical silica gel with particles size 3-10 um and pore size of 100-120A. Resolution between critical pairs corresponds to rules and specifications of USP.
Application Columns: Legacy L1 C18 HPLC column · Application compounds: Acetaminophen, Aspirin, Caffeine, benzoic acid, and salicylic acid · Mobile phase: MeOH/H2O/AcOH 28/69/3 · Detection technique: UV · Reference: USP30: NF35
Method Parameters
| Mobile Phase | MeOH/H2O/AcOH 28/69/3 |
|---|---|
| Buffer | AcOH |
| Flow Rate | 1.0 ml/min |
| Detection | UV, 270 nm |
| Class of Compounds | Drug, Acid, Hydrophobic, Ionizable |
| Analyzing Compounds | Acetaminophen, Caffeine, Aspirin, Benzoic acid, Salicylic acid |