USP Methods for the Analysis of Ibuprofen using a Legacy L1 Column
https://sielc.com/Application-USP-Methods-for-the-Analysis-of-Ibuprofen-using-a-Legacy-L1-ColumnChromatogram
Description
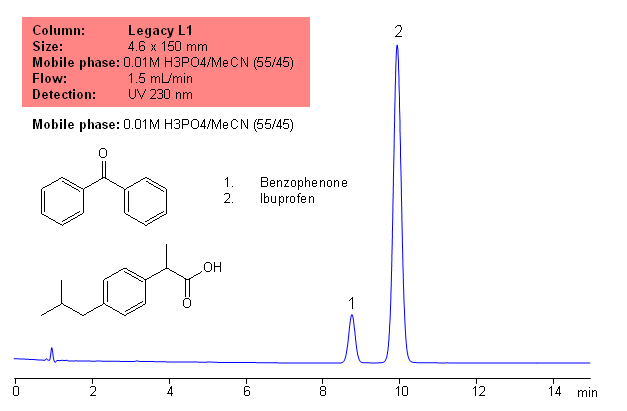
Application Notes: Ibuprofen is the most commonly used analgesic. It is a vasoconstrictor and fever reducer. According to the USP methods, ibuprofen contains no less than 97% and no more than 103% ibuprofen based on a dried basis. The USP HPLC method for the separation of procainamide was developed on Legacy L1 column according to the US Pharmacopeia methodology. L1 classification is assigned to reversed-phase HPLC column containing Ci18 ligand. Support for the material is spherical silica gel with particles size 3-10 um and pore size of 100-120A. Resolution between critical pairs corresponds to rules and specifications of USP.
Application Columns: Legacy L1 C18 HPLC column · Application compounds: Ibuprofen and benzophenone · Mobile phase: .01M H3PO4/MeCN (55/45) · Detection technique: UV · Reference: USP35: NF30
Method Parameters
| Mobile Phase | MeOH/H2O – 45/55% |
|---|---|
| Buffer | H3PO4 |
| Flow Rate | 1.5 ml/min |
| Detection | UV, 230 nm |
| Class of Compounds | Drug, Nonsteroidal anti-inflammatory drug, Hydrophobic, Ionizable, Neutral |
| Analyzing Compounds | Ibuprofen, benzophenone |