USP Methods for the Analysis of Aspirin (Acetylsalicylic acid (ASA)) Using Legacy L1 Column
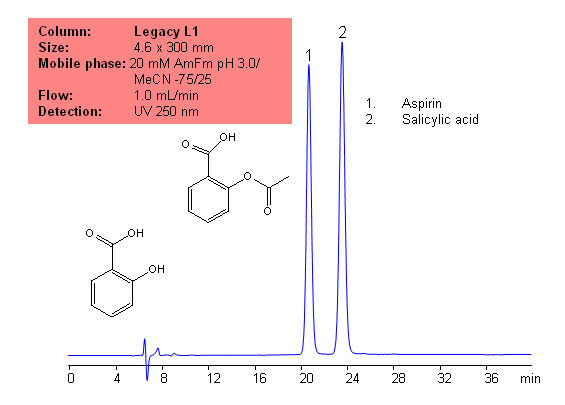
Application Notes: Aspirin is one of the oldest analgesics. While it is one of the oldests analgesics, it is still widely used today, and is still one of the most common drugs. According to the USP methods, aspirin contains not less than 99.5% and no more than 100.5 percent of aspirin calculate on a dried basis. The USP HPLC method for the separation of aspirin was developed on Legacy L1 column according to the US Pharmacopeia methodology. L1 classification is assigned to reversed-phase HPLC column containing C18 ligand. Support for the material is spherical silica gel with particles size 3-10 µm and pore size of 100-120 Å. Resolution between critical pairs corresponds to rules and specifications of USP.
Application Columns: Legacy L1 C18HPLCcolumn
Application compounds: Aspirin and salicylic acid
Mobile phase: 20 mM AmFm pH 3.0/MeCN- 75/25
Detection technique: UV
Reference: USP35: NF30
Methodenparameter
| Column | Legacy L1, 4.6×300 mm, 5 µm, 100 Å |
|---|---|
| Mobile Phase | MeCN – 25% |
| Buffer | AmFm pH 3.0 20 mM – 75% |
| Flow Rate | 1.0 mL/min |
| Detection | UV, 250 nm |